In COMSOL ®, it’s easy to set an analytic EEDF, import EEDFs, or compute the EEDF using the Boltzmann Equation, Two-Term Approximation interface. By providing electron impact cross sections, the flexibility in changing the EEDF is preserved. The reason for this is that the EEDF is not known a priori, and in low-temperature plasmas, the EEDF often deviates from a Maxwellian EEDF. For electrons, the best strategy for obtaining rate coefficients is to provide electron impact cross sections and make a suitable integration over the electron energy distribution function (EEDF). The source terms in the transport equations are computed using rate coefficients that represent the effect of collisions. In the Plasma interface and Plasma, Time Periodic interface, when defining an electron impact reaction, contributions to the source terms of the continuity equations of the species involved are added automatically, and the energy lost or gained by the electrons is added to the source term of the electron mean energy equation. This type of reactions tends to be very effective in destroying electrons with low energies. In electron attachment reactions, an electron is captured by a species, and a negative ion is created. Ionization reactions are a special case of an excitation, as an electron-ion pair is created. The inverse reaction is also possible, in which case a species de-excites and the electron gains its energy. 
In excitation reactions, an electron can create a new species with higher internal energy. 
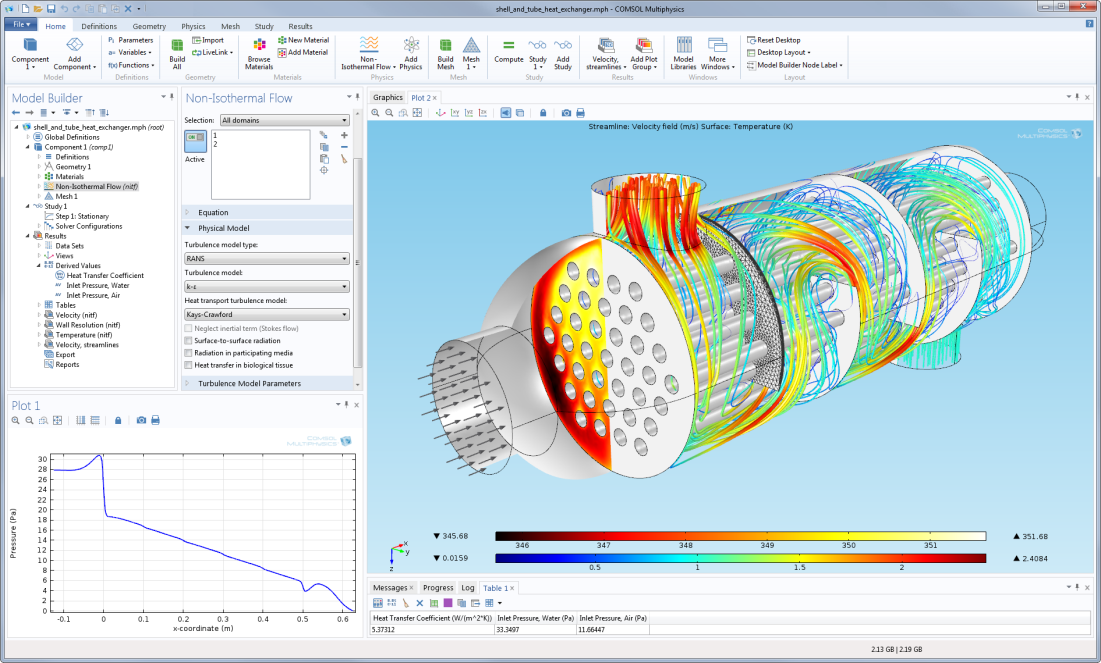
Moreover, a heating source term for the background neutral gas is created and can be coupled with the Heat Transfer in Fluids interface. When an elastic electron impact reaction is set in the Plasma interface, the electron energy loss is accounted for in the electron mean energy equation. In elastic collisions, new species are not created, but there is energy transfer between the electrons and neutrals. The reaction is specified by an electron impact cross section. The Settings window is for the Electron Impact Reaction feature that ionizes the molecular oxygen. The Model Builder showing the Electron Impact Reaction features for a user-made plasma chemistry for a mixture of argon and oxygen. You can see the settings for an ionization reaction of oxygen in the image below.įigure 1. The Electron Impact Reaction feature in the COMSOL Multiphysics ® simulation software enables you to define these types of reactions. Electron Impact ReactionsĮlectron impact reactions can be categorized as elastic, excitation, ionization, or attachment. These are discussed in more detail below. In summary, the main elements of a plasma chemistry are its species and properties, including transport coefficients, electron impact reactions, heavy species reactions, and surface reactions. For instance, it’s normal to assume that the electrons are absorbed when reaching a metal surface and that the ions are neutralized to the ground state. The interaction with the surface needs to be described. In a plasma reactor, species are transported by diffusion and migration and eventually reach a surface. Heavy species like excited states and ions can also collide, resulting in charge transfer, ionization, and ion–ion recombination. In this context, electrons can be thought of as the primary vehicle for sustaining a discharge, since they gain energy from the electric field and lose it in collisions with the background gas where the reaction products can be neutral excited states, electron, and ions. In many industrial reactors, the plasma is sustained by applying an electric field that is able to accelerate the electrons to energies where ionization can occur. For the plasmas we are interested in modeling, the electrons have much higher energies than all other species in the plasma, with the electron mean energy being of the order of a few electronvolts and the temperature of the background gas ranging from room temperature to about 1000 K. This means that the electrons and ions are transported in a background of neutral gas (with which they primarily collide). In low-temperature plasmas with low ionization degrees, the dominant species are neutrals. Workflow for Developing Plasma Chemistries. 
We will also discuss approaches to preparing plasma chemistries. Here, we present the components of a plasma chemistry as well as where and how to obtain relevant data for plasma modeling. With this information, source terms and transport coefficients appearing in the species transport equations can be computed. It’s through reactions and collisions, for example, that the interactions between different species in a plasma are specified. Plasma chemistry is of great importance to plasma modeling.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
